
Hi everyone,
Last month, we navigated FDA approvals, estradiol patch shortages, and the role of MHT in cardiometabolic health. Excitingly, we have some major clincal updates this month!
Two major bodies released or updated their guidance - appears that the frameworks are finally catching up to clinical reality! First, ACOG has formalized what many of us were already doing informally for endometriosis diagnosis. Second, FIGO has released a much-needed global framework for differentiating hormonal mood disturbances from pathological depression. Finally, we are tackling the estrogen-as-skincare conversation. Fueled by social media trends, we had a closer look at the data. As always, the gap between biological theories and clinical evidence affects how we can counsel our patients.
Here’s a quick summary of this month’s topics:
✔️ ACOG on Endometriosis: ACOG now formally supports the empiric diagnosis and treatment of endometriosis based on clinical symptom patterns, without requiring surgical confirmation. The historical 4-to-11-year diagnostic delay is explicitly named as a harm, making early, empiric medical management the new quality-of-care standard. ACOG
✔️ Estrogen & Skin (Lephart, 2026): MHT has biologically plausible effects on dermal collagen and elasticity. However, while small RCTs show modest collagen gains (+3–5% over 12 months), longer, better-powered trials (like KEEPS) show no significant change in wrinkling versus placebo. Because the data remains highly heterogeneous, there is still no indication to prescribe MHT independently for aesthetic use. Lephart, 2026
✔️ FIGO 2026 on Mood: Never rely on a PHQ-9 alone in midlife. FIGO recommends pairing it with menopause-specific tools (MRS, CIDI-VENUS) to separate hormonal mood symptoms from Major Depressive Disorder (MDD). The triage pathway is clear: transdermal estradiol is first-line for hormonal presentations; SSRIs/SNRIs when MDD is primary; and we can use both meds for mixed presentations. FIGO
Endometriosis: From Surgical Confirmation to Clinical Pattern Recognition:
ACOG's updated guidance replacing the Practice Bulletin 114 and Committee Opinion 760 is officially here, and we are shifting towards way more patient-first medicine. Historically, the framework required surgical confirmation before endometriosis could be formally diagnosed and treated. That requirement directly drove a 4-to-11-year diagnostic delay, resulting in progressive central sensitization, narrowed fertility windows, and patients arriving at specialty care having completely lost confidence in the clinicians they saw along the way.

The New Standard:
The updated guidance now formally supports presumptive clinical diagnosis and empiric medical treatment based on symptom assessment, physical examination, or both. The symptom criteria are deliberately broader, reflecting the following:
Cyclic or noncyclic chronic pelvic pain
Dysmenorrhea and dyspareunia
Dysuria and dyschezia
Infertility associated with any of the above
Clinical pearl: The explicit inclusion of noncyclic presentations and bowel/bladder symptoms (dysuria/dyschezia) is intentional, taking into account the exact symptom patterns most commonly misdirected to other diagnoses and driving the cumulative delay in care.
Imaging & Biomarkers.
Ultrasound is First-Line: Transvaginal ultrasound (TVUS) is the recommended first-line modality (Strong expert consensus, Moderate evidence). Where TVUS is not acceptable or feasible, Transabdominal ultrasound is the appropriate alternative.
Pelvic MRI should be reserved for cases where further characterization of deep endometriosis is needed to guide surgical treatment planning (Mixed expert consensus, Moderate evidence).
Given the recent commercial hype around non-invasive testing, ACOG makes a Strong expert consensus against the use of blood, urine, endometrial, or other biomarkers to diagnose endometriosis at this stage (not enough data yet on accuracy of these)..
The Evolving Role of Laparoscopy:
Diagnostic laparoscopy is no longer required before treatment begins. Yes, we heard that right. Remaining a valid option, ACOG recommends diagnostic laparoscopy is decided through shared decision-making, and can be considered even when physical exams and imaging are negative. If performed (1) a biopsy of suspected lesions is recommended for histologic confirmation; however, a negative histopathology result does not exclude the diagnosis, and (2) suspected lesions should be treated at the time of the initial laparoscopy to avoid the need for repeat procedures.
Ultimately, this update is less of a clinical revolution - it’s a formal endorsement of what careful practitioners were already doing. As a result of officially reframing a years-long diagnostic delay, the formalization matters in recalibrating patient-clinician trust.
✔️ The Clinical Takeaway: ACOG now supports empiric treatment based on clinical symptom patterns and examination alone. Diagnostic delay now has a guideline standard it can be measured against.
Estrogen and Skin Aging: What the Biology Shows, and Where the Evidence Stops.
If you’ve logged onto social media recently, you’ve probably seen the latest doc-influencer craze: applying vaginal estrogen cream on the face for anti-aging. It’s sparking a wave of questions in the clinic, notably one core issue: Does estrogen actually have meaningful effects on skin structure, and do those effects justify prescribing it purely for aesthetics?
Biologically, the mechanistic theory is sound. Estrogen receptors are abundant across most skin cell types. In the dermis, estrogen acts as the architect for the extracellular matrix, preserving collagen and elastin. However, after menopause, dermal collagen drops by approximately 2.1% per year, and skin thickness falls by about 1.1% annually. Translating to a staggering 30% loss of dermal collagen within the first five postmenopausal years, clinically, this presents as reduced turgor, barrier impairment, dryness, and delayed wound healing.
In reality, the data are mixed and highly dependent on how outcomes are measured.
The "Pro" Data: A 2023 meta-analysis (Pivazyan, ~1,500 patients) found MHT (oral, transdermal, and topical) significantly improved skin elasticity, thickness, and collagen, while reducing wrinkles. However, the included trials were small, heterogenous, and underpowered.
Conversely, the much larger, 4-year KEEPS trial found no significant difference in facial wrinkling or skin rigidity between the MHT (transdermal 17β‐estradiol and micronized progesterone or oral CEE) and placebo groups. Interestingly, KEEPS highlighted that race and ethnicity, not hormone therapy, were the strongest predictors of skin aging post-menopause. Nonetheless, the study design was flawed by relatively low MHT doses, inconsistent skin assessments, and underpowered sampling.
Moreover, observational data linking MHT to glowing, thicker skin is heavily skewed by the "healthy-user" bias.

So, does topical facial estrogen actually work?
Topical formulations target ER-β receptors (highly prevalent in the skin), allowing for localized hydration and elasticity with minimal systemic absorption. A review of 23 studies (Rzepecki, 2019) found topical estrogens generally improve dryness, texture, and wrinkles. Looking closely at the data:
Collagen: 3 months of topical estradiol boosted dermal collagen synthesis by 38% (Varila, 1995). Transdermal 17β‑estradiol increased abdominal collagen by 5.1% over 12 months (Rzepecki, 2019).
Elasticity: transdermal patch therapy use for 12 months increasedforearm skin elasticity by +3.3 percentage‑points (Rzepecki, 2019).
Thickness: 0.01% estradiol facial cream increased facial epidermal thickness by 23% in 6 months (Rzepecki, 2019).
Wrinkles: 4 weeks of estradiol gel (0.06%) improved 3D wrinkle topography in the upper limbs (Masuda, 2011).
Estriol: Even weak topical estriol thickens the papillary dermis and improves elastic fibers (Punnonen, 1987).
The Caveats: The data is very inconsistent. Variations in estrogen concentration, application methods, area of application, base formulations, and study design limit our ability to draw definitive, standardized treatment protocols.
Furthermore, iIn older cohorts, data suggests that once structural collagen degradation is established, it is partially irreversible. As Viscomi (2025) perfectly summarized: while estrogen deficiency clearly worsens skin structure, and MHT, especially upon initiation closer to the menopausal transition, often improves objective measures, the evidence remains too inconsistent to justify prescribing MHT solely for skin.
As a result of this mixed data, the regulatory and guideline consensus concludes that peer‐reviewed clinical data assessing the specific benefit–risk ratio of MHT, regarding safety and efficacy, for skin applications is limited.
If the patient is on systemic estrogen and we are addting additional topical facial estrogen, some of that topical dose could be converted into more systemic estrogen. If they have a uterus, it’s important to keep an eye on their endometrium, to make sure they are not developing a hyperplasia. Because we don't know exactly how much estrogen from facial creams enters the bloodstream, we don't know if it's safe to use without also taking progesterone to protect the uterus. Moreover, there’s been some reports of negative side effect with the use of topical estrogen on the face: pigmentation. If a patient has a history of melasma, they might be at risk of their skin darkening.
✔️ The Clinical Bottom Line: When the patient asks if estrogen cream is going to reduce their wrinkles, the honest answer is: we don’t know for sure yet. There’s not enough evidence to endorse HT formally as a treatment for skin aging. You can encourage them to try treatments that have more robust evidence like sunscreen and retinoids as first-line. And if they are not yet on systemic HT and are eligible, it’s a perfect moment to have a conversation about eligibility. Until we have more robust data, prescribing systemic MHT and topical estrogen for skin benefits remains off-label practice.
FIGO 2026: A Framework for Mental Health at the Menopausal Transition.
Up to 68% of women report heightened depressive symptoms during perimenopause, yet consolidated global guidance on how to recognize, differentiate, and treat these symptoms during menopausal transition has historically been sparse. The recent 2026 International Federation of Gynecology and Obstetrics (FIGO) best practice recommendations aim to close that gap.
The core premise of the guidelines highlights that perimenopause is a neuroendocrine transition, not just a reproductive one. Erratic fluctuations in estrogen and progesterone directly modulate serotonergic and dopaminergic pathways in the brain. Concurrently, the decline in GABAergic neurosteroids, particularly allopregnanolone, further destabilizes mood regulation. Together, these mechanisms drive the irritability, mood swings, and depressive episodes that are hallmark features of the perimenopausal years. As such, the diagnostic foundation of the new framework is separating hormonally mediated mood disturbance from Major Depressive Disorder (MDD).
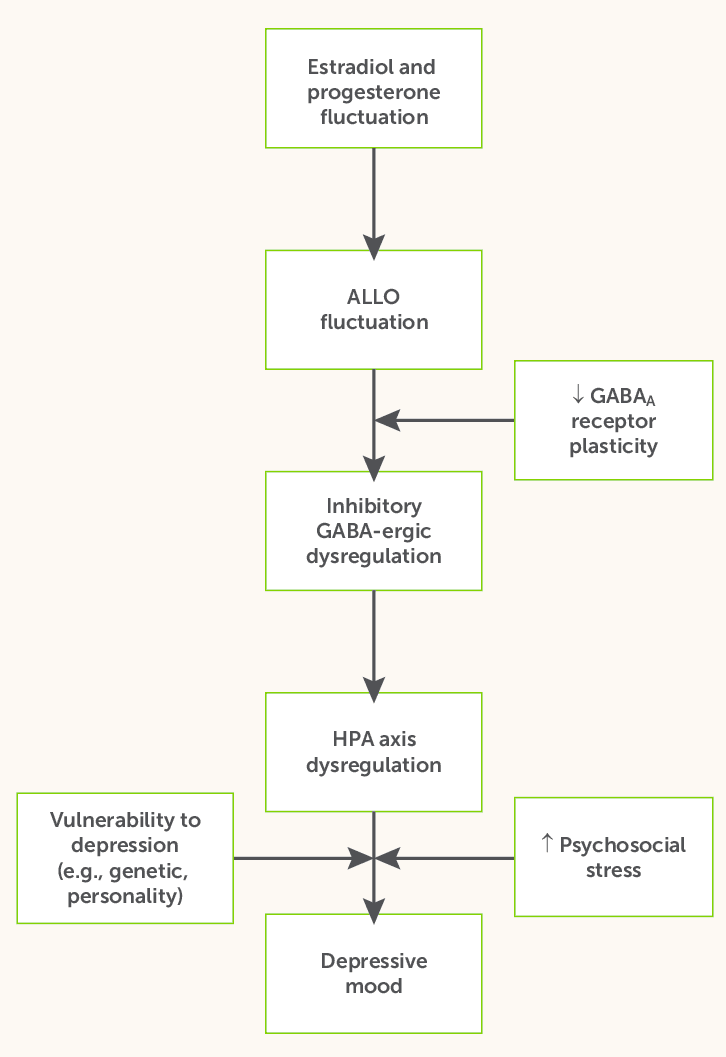
Traditionally, clinicians use the PHQ-9 to diagnose major depressive disorder, but in midlife women, the assesement cannot separate a hormonal signal from a psychiatric one. The FIGO guidelines recommend pairing the PHQ-9 with a menopause-specific tool (like the Menopause Rating Scale [MRS], MENQOL, or CIDI-VENUS) to properly triage the patient into one of three buckets:
Hormonal Mood Disturbance: Symptoms track with hormonal fluctuations and are accompanied by vasomotor symptoms (VMS) and sleep disruption. They often don’t meet full MDD criteria but severely impact quality of life. Action: Consider MHT early.
First-Line Hormonal Major Depressive Disorder (MDD): Symptoms persist independently of hormonal patterns and cause significant functional impairment. Notably, women with a prior history of MDD face a 59% recurrence risk during perimenopause, compared to just 28% for those without, a critical history-taking pearl. Action: Psychiatric treatment is required; hormone management alone is insufficient.
First-Line Psychiatric: When MDD is the primary driver, or MHT is contraindicated, venlafaxine (37.5–75 mg/day) and escitalopram (5–10 mg/day) boast the strongest efficacy data for this specific population.
Mixed Presentation: Highly common. Patients exhibit both hormonal and psychiatric features. Action: A parallel approach, initiating MHT alongside an SSRI or SNRI, is the guideline-supported strategy.
When MHT isn't an option: If mood is largely secondary to severe hot flashes and sleep disruption, and MHT isn't an option, target the VMS. Low-dose paroxetine (7.5 mg), fezolinetant, and the newly approved elinzanetant remain the only FDA-approved non-hormonal treatments specifically for VMS.
✔️ Clinical Pearl: As much as pharmacotherapy remains therapeutic for some, we cannot ignore the foundations of mental health, nor can we ignore patient preferences. In such cases, Cognitive Behavioral Therapy (CBT) carries Level 1 evidence for menopause-related insomnia and depressed mood, endorsed by both FIGO and NICE as an alternative or adjunct to pharmacotherapy. Furthermore, routine resistance training and aerobic exercise show measurable, dose-dependent reductions in perimenopausal depressive symptoms.
Conclusion
It is always worth celebrating when institutional guidelines finally formalize what practitioners are already doing in the clinics and introduce more patient-friendly guidance.
As always, if you made it to the end of this newsletter, thank you! We are thrilled you are part of this community of clinicians driving the forefront of evidence-based women's health.
Until next month,
The Dama Health Team

