We’re excited to share our January updates on key developments and research across women’s hormone health.
This month, we are bringing data to the forefront of the conversation. As conversations about menopausal hormone therapy (MHT) continue to expand, claims around the benefits beyond symptom relief, particularly around neuroprotection and dementia prevention, are circulating faster than evidence can support. Clinicians are left to navigate the space between biologic plausibility, observational findings, and what can responsibly be communicated in the exam room.
At the same time, regulatory progress continues. The FDA has recently taken a significant step towards addressing long-standing gaps in sexual health by approving flibanserin (Addyi) for the treatment of Hypoactive Sexual Desire Disorder (HSDD), in postmenopausal women under 65 years of age.
Together, these developments raise a shared question: how do we calibrate clinical expectations, language, and decision-making to align with what the evidence actually supports?
Menopause hormone therapy and dementia risk: where the evidence currently stands:
A large volume of online content currently encourages starting specific MHT formulations early in menopause for a presumed neuroprotective benefit. Much of this narrative relies on observational data, which are vulnerable to healthy-user bias, and mechanistic theories that, while compelling, remain theoretical in humans.
A recent, methodologically systematic review and meta-analysis offered a contrary conclusion to much of the current online narrative. The authors concluded that existing evidence does not reliably demonstrate either benefit or harm, including for initiation during perimenopausal years or longer duration of use.
The biological argument
efore reviewing the clinical outcomes of the meta analysis, let’s look into why the hypothesis of neuroprotection is so compelling. Much of the current optimism stems from the pioneering work of neuroscientists like Dr. Lisa Mosconi, who have built on extensive research showing that the brain is an endocrine organ densely populated with estrogen receptors, helping to elucidate how the menopausal transition affects brain metabolism. 17β-estradiol serves as a master regulator of cerebral glucose metabolism and mitochondrial adenosine triphosphate (ATP) production. Notably, Mosconi’s imaging studies have visualized a cerebral "energy crisis" during perimenopause, a bioenergetic deficit that may precede downstream pathology such as amyloid deposition.
This biological reality underlies what we know as the "critical window" hypothesis: the theory that providing estrogen during the transition might maintain neuronal fuel supply and prevent the downstream cascade of Alzheimer’s pathology. The mechanism is scientifically sound and biologically plausible; the challenge, however, has been proving that this metabolic preservation translates into long-term clinical dementia prevention in human trials.
What was reviewed, and why it matters.
Against this backdrop, the meta-analysis reached a different conclusion. The review included 15 studies encompassing more than one million women and examined outcomes including all-cause dementia, Alzheimer’s disease, and combined mild cognitive impairment (MCI) across a range of systemic and local MHT regimens.
Only one randomized trial was available (WHIMS), which evaluated women initiating standard‑dose oral conjugated equine estrogens (CEE)‑based regimens at age 65 or older. Although the trial showed a relative increase in dementia risk, the absolute difference was small, approximately 6–7 additional cases per 1,000 women, with wide confidence intervals. Due to the largely outdated CEE + medroxyprogesterone acetate (MPA) regimens and initiation occurring much later than is currently recommended, these findings have limited applicability to modern midlife HT prescribing but serve as a clear safety signal for late initiation.
Across observational studies, estimates for estrogen-only and combined therapy generally showed little to no difference in dementia risk compared with non-use and were graded as very low certainty. Where signals suggested possible harm with late initiation or combined therapy, findings were inconsistent and highly sensitive to study design limitations and residual confounding.
Crucially, analyses stratified by timing of initiation did not confirm the "critical window" hypothesis for dementia prevention. Results remained underpowered with wide confidence intervals, meaning we cannot claim that early initiation prevents cognitive decline based on current data.

Where this leaves us:
The largely neutral findings should not be viewed negatively; rather, they reflect genuine uncertainty. The absence of high-quality evidence for neuroprotection does not negate MHT's value. And as per usual with research, it often just guides us towards further research that needs to be done.
Perhaps long-term RCTs starting in midlife are unlikely, but progress can be achieved with high-quality real-world datasets and well-designed observational studies with careful phenotyping, integration of physiological biomarkers, and more granular hormone exposure data. Advancing this kind of evidence generation is a central focus of the work we are building toward at Dama Health.
Practical implications for clinicians
✔️ Current evidence does not support MHT being prescribed for dementia prevention at any age. This aligns with guidance from NICE, the USPSTF, and the Lancet Commission.
✔️ There is biological plausibility of MHT supporting brain health; however, we need to remain honest and transparent about the level of evidence.
✔️ Be transparent that while it may help clear the brain fog driven by hot flashes and sleep loss, it has not been proven to protect the brain from aging.
Menopause, MHT, and the Brain: What Large-Scale Data Actually Show.
Patients frequently report "brain fog," mood changes, and sleep disruption during (peri)menopause. Because these symptoms feel neurological, both clinicians and patients hope that hormone therapy might offer structural brain protection.
A large UK Biobank analysis provides one of the most comprehensive looks to date at how menopause and MHT intersect with mood, cognition, sleep, and brain structure. The findings are measured, but instructive.
What the data show:
Across nearly 125,000 women, postmenopause was associated with higher rates of depression, anxiety, insomnia, and tiredness compared with premenopause. Women using MHT also reported a greater symptom burden than postmenopausal non-users, a pattern most consistent with confounding by indication, whereby women with more severe or pre-existing psychological symptoms were more likely to receive therapy. Absolute differences in symptom scores and sleep duration were modest, with small effect sizes despite statistical significance.
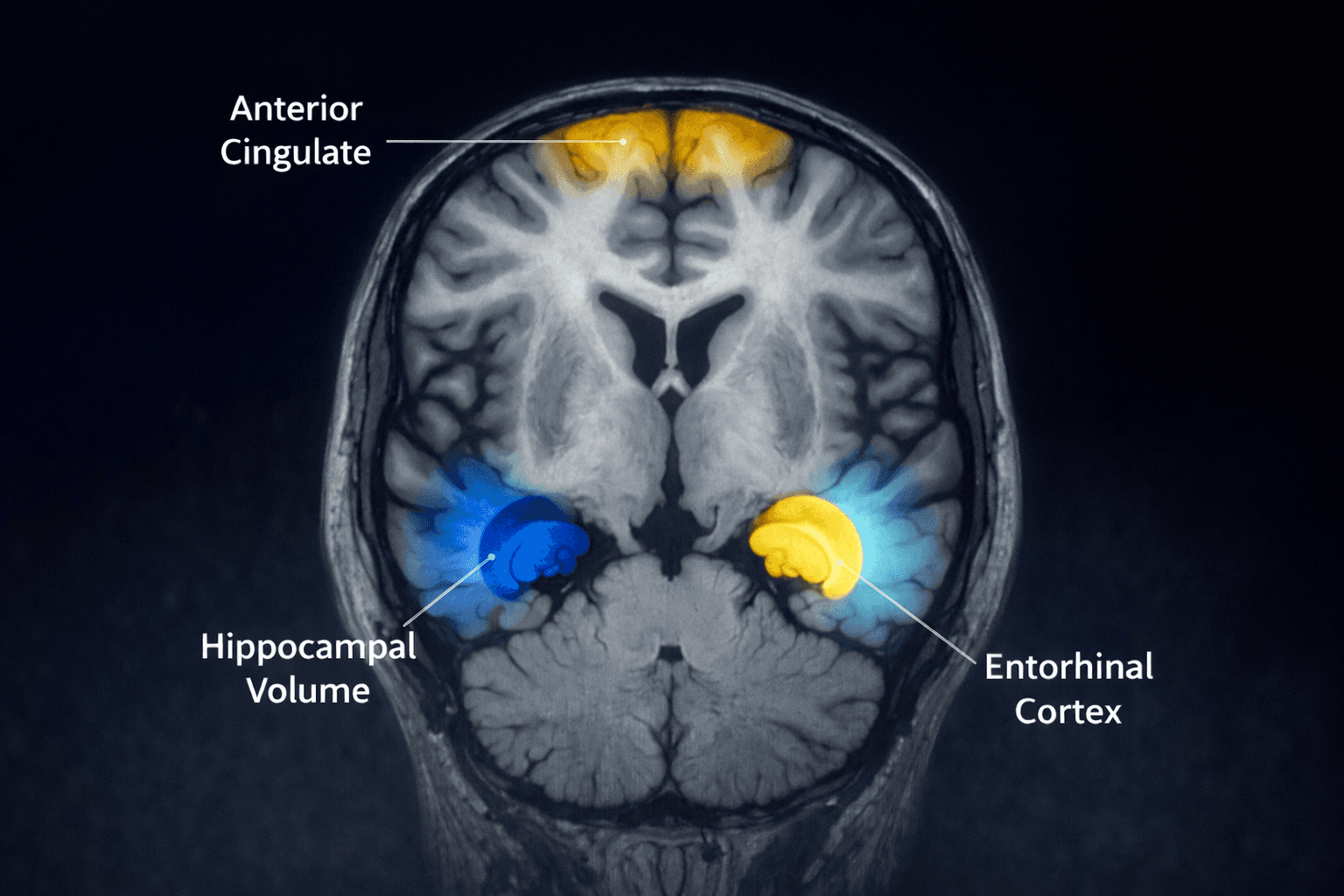
MHT users also showed slightly smaller gray matter volumes in select regions involved in memory, emotion, and decision-making. These differences were biologically small, with menopause stage and MHT status together explained ≤0.5% of the variance in hippocampal volume. Additionally, cognitive task performance was similar across groups, and no consistent memory benefit of MHT was observed. Clinically, these findings support screening for and treatment of mood and sleep disorders during the menopausal transition rather than the use of MHT as a stand-alone intervention for psychological symptoms.
Interpretation is limited by single time-point brain imaging, self-reported hormone use, inferred duration of use, and lack of formulation-specific analyses. Much of the UK Biobank exposure likely reflects older oral CEE + MPA regimens, which is relevant because MPA can attenuate estradiol’s neuroprotective effects, and standard-dose oral CEE, particularly when combined with MPA, is known to increase hepatic inflammatory and prothrombotic markers (1,2).
Notably, reduced hippocampal volumes have been most consistently observed with late initiation or prolonged use (>10 years postmenopause or after age 65), whereas early initiation shows a protective pattern in select populations, including women with surgical menopause or APOE4 carriers (3,4). Reassuringly, long-term follow-up from KEEPS suggests that short-to-medium–term MHT use in early menopause is not associated with delayed cognitive harm (5)
How to use this clinically:
This study is well-powered to detect associations between menopause stage, symptom burden, and healthcare use, but not to determine which hormone therapies may help or harm which patients. The broader takeaway is that MHT is not a panacea for menopausal mood or cognitive disorders. The "brain fog" of menopause is multifactorial, driven by sleep fragmentation, psychosocial stress, and metabolic changes as much as by estrogen withdrawal.
Clinical takeaways:
✔️ Expect mood and sleep symptoms across the menopausal transition, regardless of MHT use.
✔️ Do not frame MHT as a stand-alone treatment for primary depression or anxiety. If mood symptoms persist despite adequate estrogen, screen for primary psychiatric disorders and treat accordingly.
Expanding Sexual Health Options: What Actually Changes in Practice
The FDA, yet again, has demonstrated increasing regulatory attention to women’s health care gaps, expanding approval of flibanserin beyond premenopausal women, and to postmenopausal patients with hypoactive sexual desire disorder (HSDD). Profoundly undertreated, HSDD is defined as persistent low sexual desire causing distress, not better explained by another medical or psychiatric condition. This makes Addyi the first FDA-approved pharmacologic therapy for HSDD across this broader age range.
Flibanserin is a non-hormonal, centrally acting agent that modulates neurotransmitters via serotonergic, dopaminergic, and noradrenergic pathways involved in sexual motivation, rather than “restoring libido” or addressing root relational, psychological, hormonal, or contextual drivers of sexual desire.
This approval is a victory for patient access and autonomy, validating HSDD as a treatable biological condition. However, clinical application requires nuance. Because of the daily dosing requirement, delayed onset of action (4–8 weeks), and known tolerability profile (dizziness, somnolence, hypotension, and alcohol interaction), the benefit is often incremental and highly patient-specific.
Clinical pearls:
✔️ Consider flibanserin when distressing low desire persists after addressing the "low hanging fruit": Genitourinary Syndrome of Menopause (GSM), dyspareunia, mood disorders, sleep fragmentation, medication side effects (SSRI/SNRIs), and relationship context. Consider flibanserin only after a careful biopsychosocial assessment of desire, distress, and contributing factors.
✔️ Safety First: Careful screening for hypotension risk and alcohol use is essential. While the strict REMS alcohol ban was modified in 2019, patients must still be counseled to wait at least two hours after consuming alcohol before taking their bedtime dose to minimize syncope risk.
✔️ Counsel on “The Why”: Explain to patients that this medication targets the brain’s chemistry of desire, not their hormones. It is not an "on-demand" aphrodisiac but a daily maintenance therapy. Set expectations clearly: modest effect size, daily dosing, and the need for reassessment.

Conclusion
In women’s hormone care, progress doesn't always look like discovering a new mechanism and a new drug. Sometimes, it looks like refining the boundaries of the methods we already have, clarifying where they are effective and where they are not.
In summary, the current evidence shows us that MHT does not offer a guaranteed shield against dementia, nor a simple fix for the complex neurobiology of menopausal mood.
Our role is to offer a menu of options to the patient, with pros and cons and transparency about what we know and what we don’t know just yet. We can offer a thoughtful discussion of hormonal and non-hormonal options (like the newly expanded indication for Addyi) and acknowledge their desire for prevention and neuroprotection - there are so many impactful and meaningful lifestyle changes one can implement for that.
Warmly,
The Dama Health Team

