
In October 2025, the FDA announced the removal of the long-standing boxed warning from estrogen-containing therapies. Six months later, we’re seeing the real-world effects:
a surge in demand for transdermal estradiol,
patch shortages,
and renewed public conversation about menopausal hormone therapy (MHT).
More recently, the FDA approved label revisions for 6 of 29 MHT products across four therapy categories following a comprehensive review of the evidence on cardiovascular disease, breast cancer, and probable dementia.
In this month’s newsletter, we’ll explore how MHT risks are being communicated, how supply limitations shape prescribing in practice, and summarize new real-world data examining MHT use in women with cardiometabolic disease, specifically MASLD.
Here’s a quick summary of this month’s topics:
✔️ The FDA approved labeling changes for six menopausal hormone therapy products, removing certain cardiovascular disease, breast cancer, and probable dementia statements from the boxed warning. This is a communication shift—not a universal risk reset. (U.S. Food and Drug Administration)
✔️ Estradiol patch shortages are real and granular (brand/strength/package-specific), and the near-term friction is likely to continue.
✔️ A new large real-world analysis (TriNetX target-trial emulation) shared in the materials below suggests lower major adverse liver outcomes (MALO) and modest cardiometabolic benefit among peri-menopausal women with MASLD receiving HRT, biologically plausible, clinically intriguing, and still observational (MASLD).
The FDA Removed the MHT Boxed Warnings. Here is the Real Clinical Translation:
On February 12, 2026, the FDA made a historic update to menopausal hormone therapy (MHT) labeling, approving the removal of the boxed warnings for cardiovascular disease, breast cancer, and probable dementia across six specific products (U.S. Food and Drug Administration).
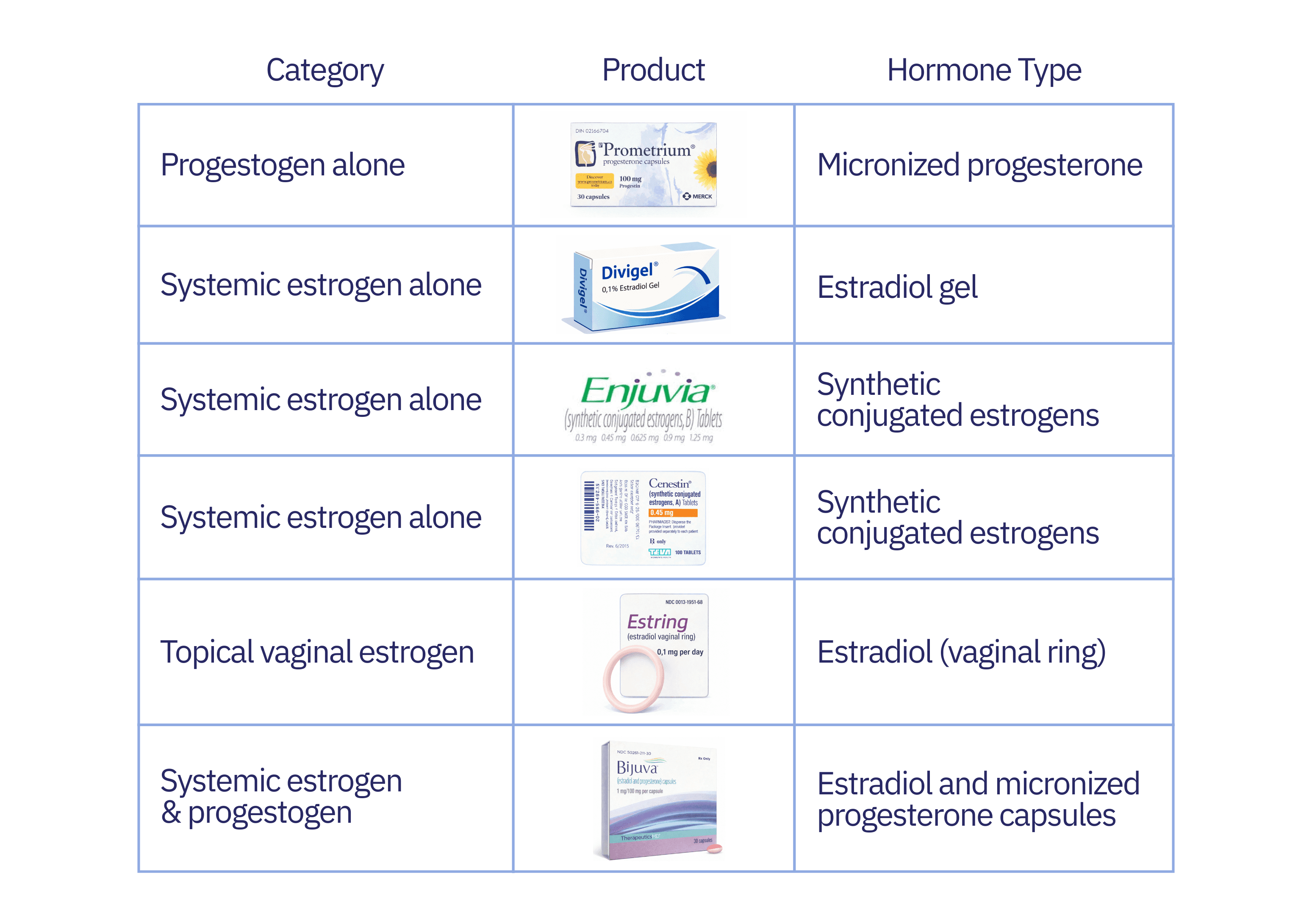
Progestogen alone: Prometrium
Topical vaginal estrogen: Estring
Systemic estrogen and progestogen: Bijuva
It’s worth noting that these products vary in the exact formulation and are not pharmacologically interchangeable (Table 1). 4 of these 6 MHT products consist of “bioidentical” formulations.
Table 1.
While the media may translate this removal to "the risk is gone," as clinicians, our job is to understand the nuance.
Here is where the data actually stands on the "Big Three" outcomes, translated into absolute terms for your patient counseling.
Cardiovascular disease: neutral when started early.
Much of the public narrative relies on relative risk framing (e.g., “50% lower cardiovascular disease if started within 10 years of menopause”), but absolute effects are small. In the Cochrane review, cardiovascular events occurred in 1.8% of placebo users versus 1.1% of oral MHT users — an absolute difference of 0.7%.
In WHI, combined CEE+MPA showed no reduction in coronary heart disease, while estrogen-only therapy in women aged 50–59 translated to approximately 11 fewer cases per 10,000 women per year (0.11% annually), derived from subgroup analysis.
Surrogate trials such as ELITE showed statistically significant slowing of carotid intima-media thickness progression (–0.0033 mm/year), statistically significant but substantially smaller than statin-associated reductions (~–0.0145 mm/year), without demonstrated cardiovascular event reduction.
The Clinical Bottom Line: For healthy women under 60 or within 10 years of menopause, MHT is safe and cardiovascularly neutral. However, we don’t have robust data showing that it could be used as a primary prevention strategy for cardiovascular disease.
Dementia: Early Neutrality, Late Harm.
Cognitive data are frequently overstated in the public domain. While observational studies (like the CLSA) often make headlines, they are still hypothesis-generating studies.
Here’s what the data from randomized controlled trials is showing:
Early Initiation (Neutrality): The KEEPS trial randomized women within ~3 years of menopause to oral CEE (0.45 mg) or transdermal estradiol (50 mcg), with cyclic micronized progesterone. Over four years, and at a 10-year follow-up, there was no cognitive benefit, but also no harm. MRI markers of white matter hyperintensities and infarcts remained unchanged compared to placebo.
Conversely, the WHIMS trial demonstrated that starting combined oral CEE+MPA after age 65 carried measurable harm (approx. 1 excess dementia case per 436 treated women).
Late Initiation (Harm): The WHIMS trial studied MHT initiation at age ≥65 and demonstrated measurable harm with combined oral CEE+MPA, resulting in approximately 1 excess dementia case per 436 treated women.
Estrogen Alone (Nuance): Extended WHI follow-up suggested a 26% relative reduction in Alzheimer’s mortality with CEE alone, but the absolute effect was small - requiring roughly 2,000 women to be treated to prevent a single Alzheimer’s death.
The Brain Health Horizon: Pioneering research from Dr. Lisa Mosconi and others reveals that perimenopause drastically alters brain bioenergetics. This suggests there might be a narrow perimenopausal window for neuroprotection, but until precision-medicine trials prove this, MHT is not a preventative tool.
Clinical interpretation: Currently we know that early MHT is cognitively neutral. Late initiation of oral CEE+MPA carries measurable harm. There is currently no high-quality evidence to support prescribing MHT as a dementia-prevention strategy.
Breast cancer: regimen and formulation matter:
The Data (WHI): Combined oral CEE + MPA increased invasive breast cancer by about 8 additional cases per 10,000 women per year. In stark contrast, estrogen-only CEE in hysterectomized women actually reduced invasive breast cancer by about 7 to 10 cases per 10,000 women per year and was associated with lower breast cancer mortality on extended follow-up of 18 years.
The Data (Observational): The French E3N cohort (80,377 postmenopausal women with a mean follow-up 8.1 years and 2,354 invasive cancers) strongly suggests that the progestogen matters. Estradiol combined with synthetic progestins was associated with an increased breast cancer risk (69% higher risk versus never use), corresponding roughly to going from about 8 cases per 1,000 women over 8 years to about 14 per 1,000. Whereas estradiol combined with micronized progesterone showed no increased risk over 8 years.
The Clinical Bottom Line: Combined synthetic progestin regimens appear to slightly increase breast cancer risk over time. Estrogen-alone reduces risk in hysterectomized women. While randomized comparative data are lacking, observational data strongly favor micronized progesterone for breast safety.

The Final Takeaway for Your Practice
The removal of the FDA's boxed warnings is a massive win for shared decision-making, allowing us to counsel based on science rather than fear. However, as headlines shift public perception, patients may understandably misinterpret this update to mean MHT is a risk-free "fountain of youth."
We can ground these conversations in evidence. This labeling change simply validates what menopause specialists have known for years: MHT is highly effective, and absolute risks are incredibly low for the right candidate. Yet, the core tenets of safe prescribing remain exactly the same:
The Indication: We prescribe MHT primarily for symptomatic relief and bone protection, not as a blanket preventative therapy for chronic disease. If anything changes, we will let you know!
The Precision: Timing, route, formulation, and the patient's baseline risk are still the crucial variables that dictate safety.
The Standard of Care: Thorough shared decision-making and clearly documented, individualized benefit-risk discussions are as important as ever.
Estradiol Patch Shortages: Navigating Supply Constraints.
Transdermal estradiol has become the default "safer" route for many clinicians, particularly for patients with elevated triglycerides, VTE concerns, or metabolic vulnerability. However, following the FDA labeling update, surging demand has exacerbated existing patch shortages. As of mid-February 2026, ASHP reports rolling backorders across several manufacturers (including Sandoz and Amneal).
For clinicians, this shortage means managing symptom rebound, adherence drop-offs, and the clinical headache of forced route switches that alter a patient's risk profile.
To avoid the pitfalls of assuming patches are perfectly interchangeable or defaulting to oral estradiol without revisiting safety, here is a practical substitution framework:
A practical substitution framework:
If a patient cannot obtain her usual estradiol patch, consider the following framework:
1. Stay transdermal if possible (Patch-to-Patch)
Switch to an equivalent total weekly estradiol dose using a different brand or frequency schedule. Clinical pearl: Be explicit about application timing if moving a patient from a once-weekly to a twice-weekly system, and re-review adhesion troubleshooting.
2. Pivot to gels or sprays (transdermal).
Gels (e.g., Divigel, EstroGel) and sprays (Evamist) maintain the critical benefit of bypassing first-pass hepatic metabolism and are often easier to source during patch shortages. Clinical pearl: Re-counsel on application sites, required drying times, and strict precautions against skin-to-skin transfer to pets or family members.
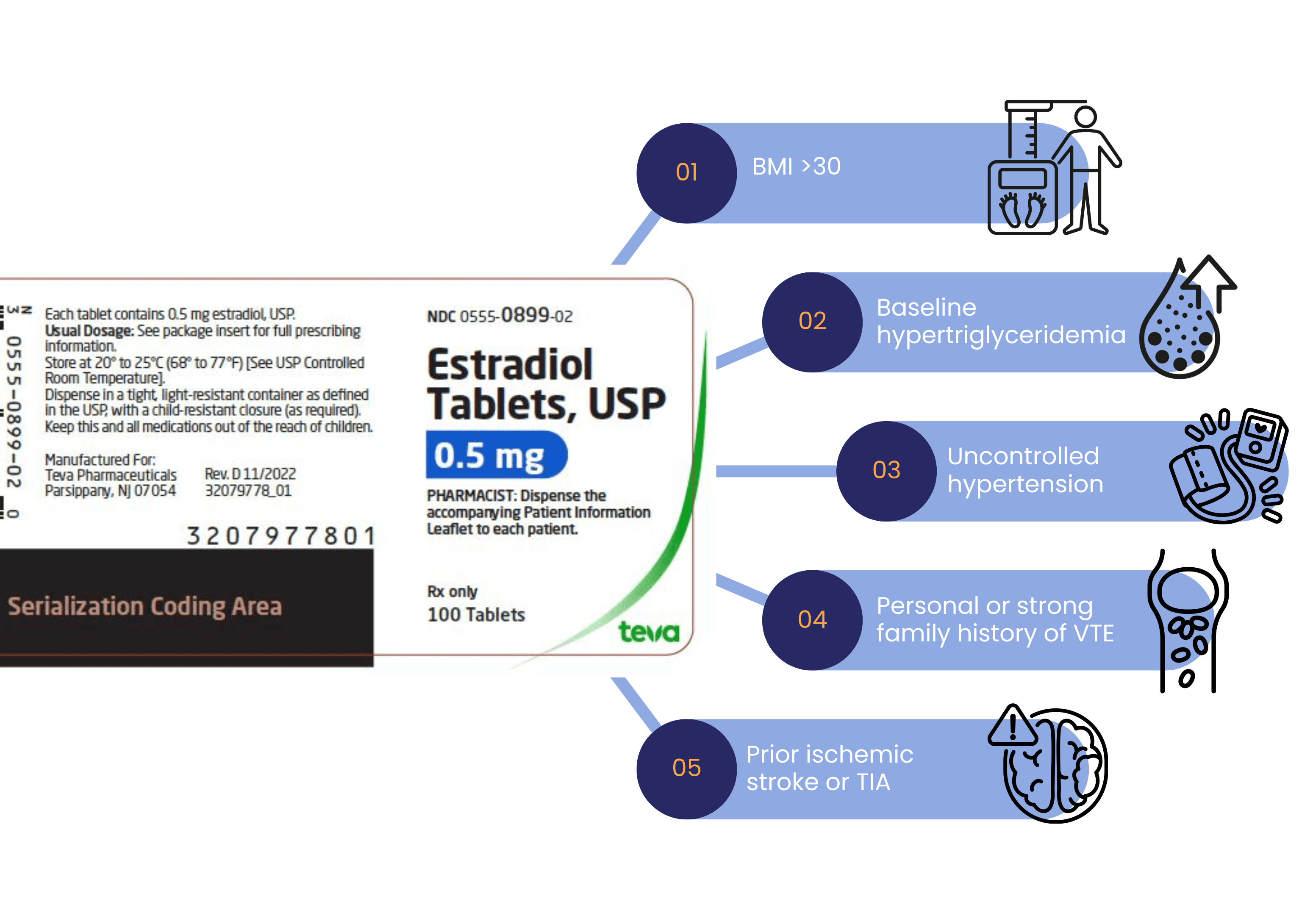
3. If moving to oral estradiol, re-stratify risk
Oral estradiol introduces first-pass hepatic effects, modest triglyceride shifts, and small prothrombotic changes. While absolute risks remain low for healthy, early postmenopausal women, you must re-anchor the safety conversation. Pause and reassess for:
Hormone Therapy and fatty liver in midlife: what this new “target trial” study found:
Metabolic dysfunction-associated steatotic liver disease (MASLD) intersects heavily with the menopausal transition. A recent real-world “target trial emulation” using the TriNetX EHR database asked a highly pragmatic question:
If a perimenopausal woman with MASLD starts hormone therapy, does it reduce her risk of serious liver outcomes?
Looking at a matched cohort of over 43,000 women (ages 40–65) over five years, the data revealed small but measurable absolute benefits for hormone therpay users.
The Findings (Absolute Numbers):
Severe Liver Events: 0.57% absolute risk reduction (translating to 5–6 fewer serious events—like cirrhosis or ascites—per 1,000 women treated over 5 years)
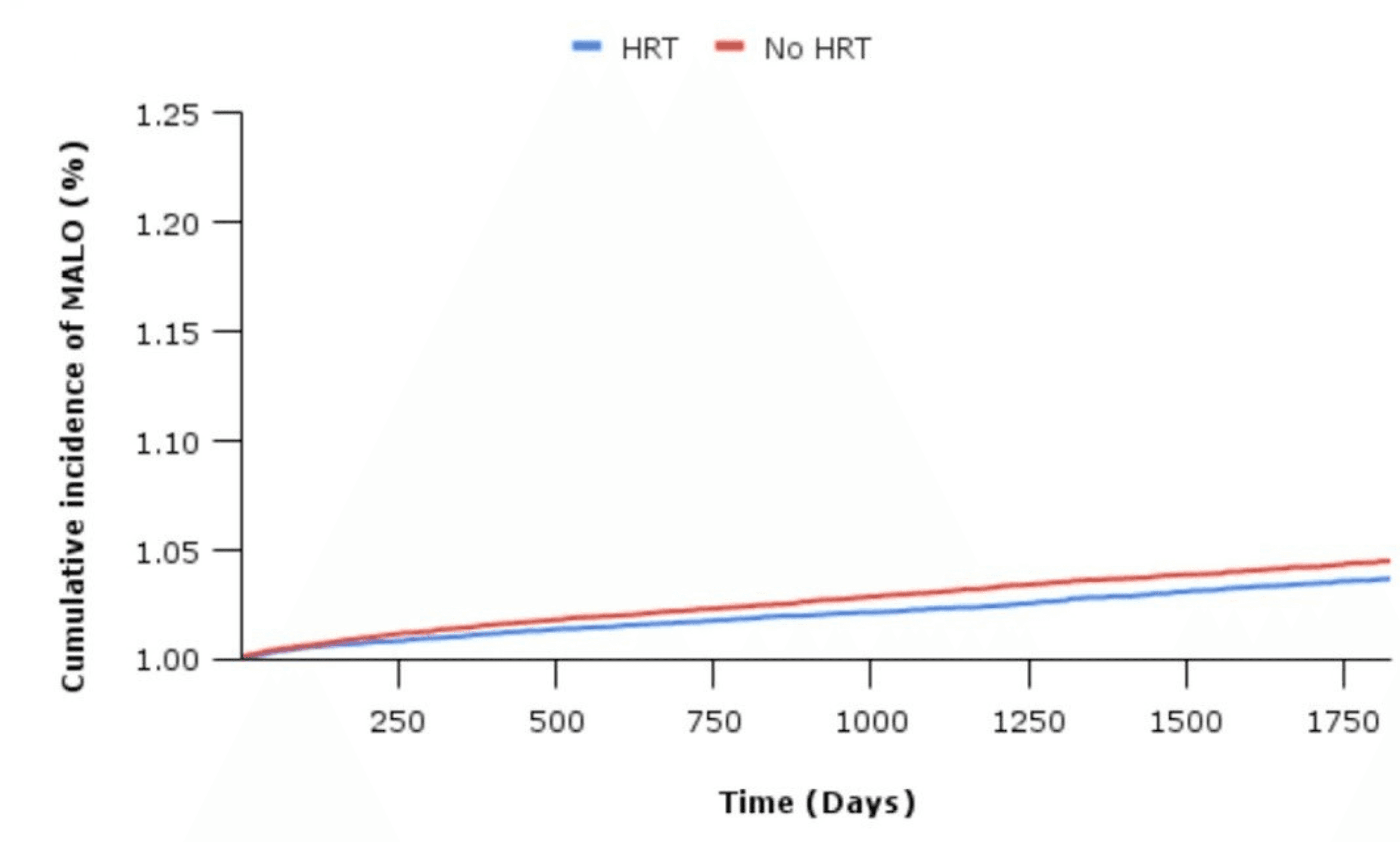
Incident Type 2 Diabetes: 1.28% absolute reduction (~13 fewer cases per 1,000 women) for women using hormone therapy.
Major CVD Events: 0.48% absolute reduction (~5 fewer events per 1,000 women). This is potentially clinically meaningful given that this population is at high-risk.
Where Interpretation requires restraint before framing HT as a liver-protective therapy, we have to acknowledge the limits of EHR data:
No route stratification: "HRT" was defined broadly. The dataset couldn't separate oral from transdermal users, which is a massive clinical limitation when discussing liver pathology.
Imperfect staging: Fibrosis stage—the strongest predictor of liver outcomes—was approximated using labs and proxies rather than uniform histologic staging.
Residual confounding: It is an observational emulation, not an RCT.
The Clinical Bottom Line: This study does not override guideline-directed MASLD pharmacotherapy, nor does it make MHT a primary treatment for liver disease or diabetes. However, it provides a highly reassuring context: If your MASLD patient needs MHT for menopausal symptoms, there appears to be a secondary metabolic and hepatic upside.
Clinical Pearl: In patients with MASLD or broader metabolic vulnerability, transdermal estradiol remains the vastly superior route to bypass first-pass hepatic metabolism, avoid triglyceride elevations, and minimize prothrombotic risk.
Conclusion
In women’s hormone care, progress is often less about a brand-new therapy and more about what we say out loud, what we put in labels, and what patients can actually access on a Tuesday afternoon.
This month’s updates show exactly what the maturation of menopause care looks like:
Retiring fear: The FDA’s warning removal allows us to move past fear-based prescribing without abandoning careful risk stratification.
Managing access: The estradiol patch shortages remind us that continuity planning is no longer an administrative afterthought and that we need to be up to date with alternative products and estrogen dose equivalents.
Widening the lens: The MASLD data adds to the body of evidence that the menopausal transition is a critical cardiometabolic inflection point, not just a reproductive health transition.
If you got to the end of this newsletter - thank you for reading and being part of this community of clinicians who are on the forefront of evidence-based women's health!
Warmly,
The Dama Health Team

