
What a year for (peri)menopause! 2025 marked a real shift - not just in visibility, but in momentum. We saw menopause move further out of the shadows and into serious conversation: more rigorous research in women’s hormone health, growing recognition from employers supporting midlife women, and meaningful policy signals (including the FDA’s removal of the black box warning). This work is just getting started. We’d love to see more research and industry collaborations - progress will require sustained, multi-year collaboration between innovators, regulators, pharma, and payers.
As we head into the new year, there’s genuine cause for cautious optimism: new therapeutic candidates are emerging, assumptions are being questioned, and the clinical conversation is becoming more nuanced.
TL;DR
✔️ Hot-flash–targeted non-hormonal therapies are increasingly differentiated not by efficacy alone, but by sleep effects, tolerability, and monitoring burden.
✔️ Early pharmacokinetic data on estradiol-only implants raise important questions, though evidence remains preliminary and interpretation requires caution.
✔️ Persistent limitations in estradiol assays continue to shape clinical decision-making more than we often acknowledge, particularly post-menopause.
Hot flashes: alternative symptom-specific treatments:
With the recent approval of elinzanetant, clinicians now have two FDA-approved non-hormonal options for vasomotor symptoms: fezolinetant (approved in 2023) and elinzanetant.
Both target the same underlying problem: as estrogen declines, the brain’s temperature control system becomes overly sensitive (kisspeptin-specific hypothalamic thermoregulatory instability to be precise).
Fezolinetant blocks the NK3 receptor, preventing neurokinin B from overstimulating thermoregulatory neurons. Elinzanetant blocks both NK3 and NK1 receptors, dampening not only heat-trigger signals but also substance P - mediated arousal pathways, which likely explains its more consistent effects on sleep maintenance, decreasing sympathetic arousal and nighttime awakenings.
If fezolinetant turns down the heat signal, elinzanetant also turns down the signals that keep patients awake.
So, how do they compare?
There are no head-to-head trials, and indirect comparisons should be interpreted cautiously given differences in trial design, baseline symptom severity, BMI, and outcome measures. That said, a systematic review compared the two drugs:
✔️ Both agents reduce hot flash frequency by ~2–3 episodes/day at 12 weeks.
✔️ Fezolinetant shows a clear dose–response effect.
✔️ Elinzanetant shows a modest additional reduction in VMS frequency and a meaningfully larger improvement in sleep-related outcomes.
Sleep is not a “secondary” endpoint for many patients, it’s the symptom that determines quality of life.
Safety and monitoring: this matters in real-world care.
Fezolinetant carries a small but real risk of reversible liver enzyme elevations, requiring routine LFT monitoring.
Elinzanetant has not shown a comparable hepatotoxicity signal to date, though exposure numbers remain smaller.
Drug-related adverse events (including headache) were lower with elinzanetant vs placebo.

What’s next in the pipeline:
Ongoing trials are now exploring elinzanetant’s role in sleep disturbance (sleep as primary outcome) and its use alongside hormone therapy, including in women with or at high risk for breast cancer.
Zooming out: where this fits in menopause care:
Having more treatment options can only be a good thing. Novel non-hormonal therapies are being developed both as alternatives to MHT and as adjuncts for additional symptom control.
That said, it’s important to be clear: non-hormonal agents expand our options, but they do not replace hormone therapy for women who are appropriate candidates. When prescribed thoughtfully, MHT remains the most effective way to address the underlying hormonal decline driving menopausal symptoms for patients who are eligible.
Clinical takeaway:
✔️ Non-hormonal VMS therapies are not interchangeable.
✔️ Sleep effects, headache risk, and liver monitoring burden are currently the key differentiators for consideration when prescribing.
✔️ These agents are particularly relevant for women who cannot use, have contraindications to, or prefer not to use MHT, as well as for those requiring adjunctive symptom management.
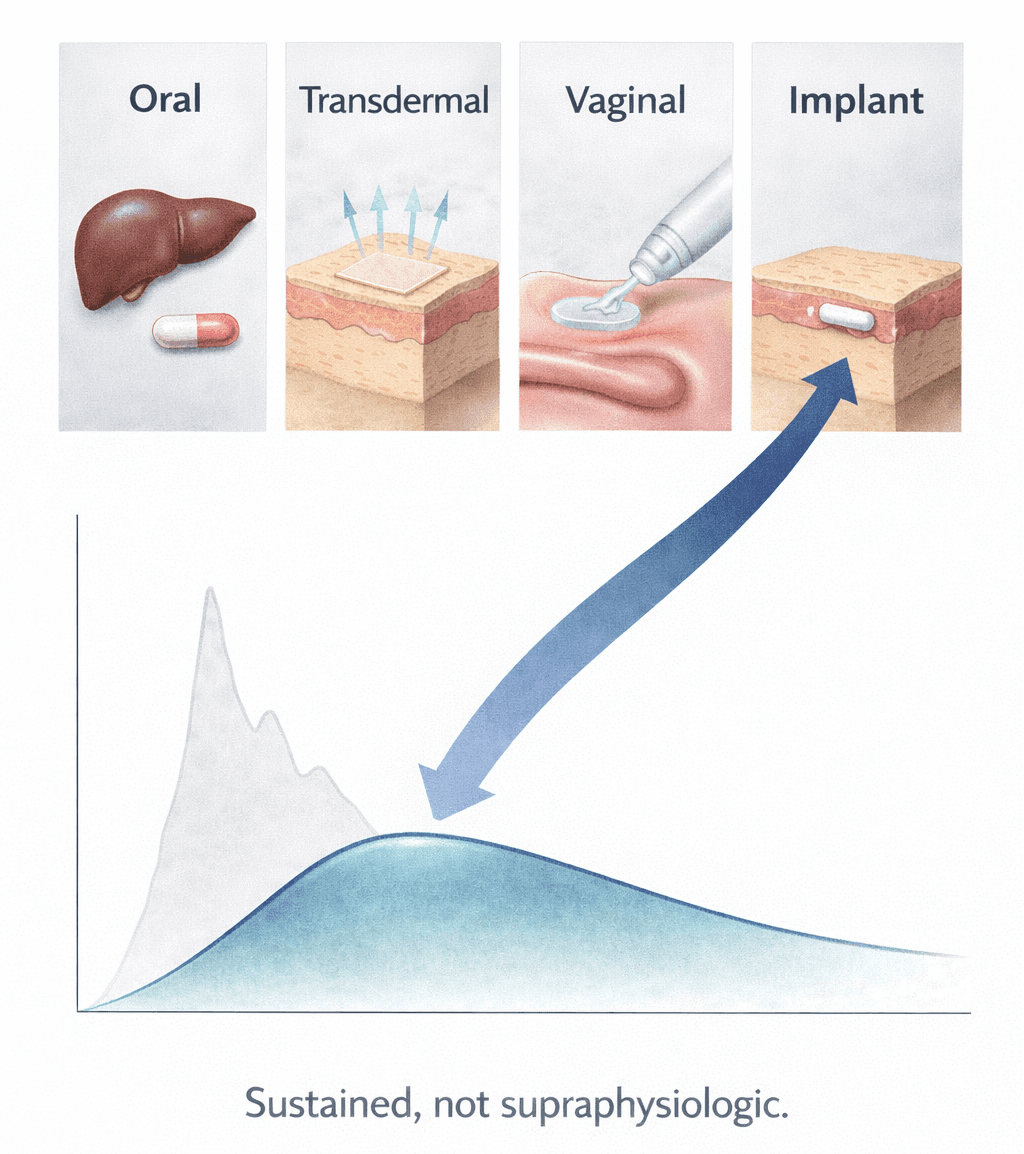
Revisiting estradiol-only bioabsorbable implants.
Estrogen-only therapy is typically reserved for postmenopausal women who have undergone a hysterectomy. Today, FDA-approved systemic options remain limited to oral and transdermal formulations, each with tradeoffs clinicians know well. Oral estrogens introduce first-pass hepatic and metabolic effects; transdermal delivery can be limited by skin tolerability and variable absorption. Vaginal estrogen, while highly effective locally, does not provide meaningful systemic exposure.
Estradiol pallets have long sat outside mainstream practice. They lack FDA approval largely due to historical concerns around pharmacokinetic unpredictability, prolonged or supraphysiologic exposure, and limited high-quality safety data. A small pilot study published this month revisited these concerns using contemporary pharmacokinetic methods.
In this study, 20 healthy hysterectomized women (≤59 years) with menopausal symptoms and biochemical hypoestrogenism received a 25-mg subcutaneous estradiol implant and were followed for 24 weeks. Estradiol levels rose gradually within days and remained sustained over the study period. Estradiol-to-estrone ratios stayed stable, and average circulating concentrations were modest, though interindividual variability was considerable, driven by differences in release kinetics rather than estradiol metabolism.
From a clinical perspective, symptoms improved: overall Menopause Rating Scale scores fell by ~60%, severe hot flashes dropped from 60% to 5%, and no serious safety signals emerged over 24 weeks, aside from mild-to-moderate headaches and breast tenderness.
Clinical Pearl:
This study doesn’t mean pallets will now be recommended for routine use, but it challenges the assumption that all implant-based estradiol delivery inevitably leads to supraphysiologic exposure.
Clinical takeaway:
✔️ Modern pharmacokinetic approaches may allow a more rigorous re-evaluation of estradiol implants (e.g. pallets!).
✔️ If replicated at scale, implants could eventually represent an additional method for systemic estrogen delivery in carefully selected patients, but we’re not there quite yet.
Why estradiol measurement still limits care
Accurate measurement of circulating estradiol in blood (serum/plasma) remains technically challenging in postmenopausal women. At low female-range concentrations, commonly used immunoassays lack sensitivity and specificity, frequently overestimating estradiol levels and showing wide inter-laboratory variability. These limitations become even more pronounced in women using exogenous hormones or aromatase inhibitors.
Liquid chromatography–tandem mass spectrometry (LC-MS/MS), the method used in the pharmacokinetic study above, offers far greater analytical specificity and is currently the only CDC-certified approach for serum estradiol measurement. It aligns more closely with reference values, especially at low concentrations where immunoassays are least reliable. However, MS-based assays require specialised infrastructure and expertise and remain far less accessible than automated immunoassays in routine care.
As a result, it’s difficult to guide MHT prescribing and dosing optimization based on serum hormone levels. Meaningful progress will depend on broader access to MS-based serum testing, continued CDC-led standardization efforts, and the development of clinically meaningful reference ranges for menopausal women.
Clinical Pearl:
At very low hormone concentrations, how you measure often matters more than what number you get.
Clinical Takeaway:
✔️ Low serum estradiol values should be interpreted cautiously, particularly when laboratory results and clinical symptoms don’t align.
Clinical Takeaway:
Well, that was a pharmacology-heavy edition! As menopause care continues to evolve, understanding the nuance behind new therapies, delivery systems, and measurement limitations matters more than ever.
Thank you for reading and for being part of this growing conversation around women’s precision hormone health.
Warmly,
The Dama Health Team

